Our editor-in-chief, Dr. Damon Adams, interviews Dr. Howard Glazer—an experienced practitioner, international lecturer, and one of Dentistry Today’s Leaders in Continuing Education—on a variety of hot topics dealing with dental materials, products, technologies, and other clinically related matters.
What has changed since your last article [Dentistry Today, June 2015]? Have some of your thoughts related to the future of materials, products, and technologies come true?
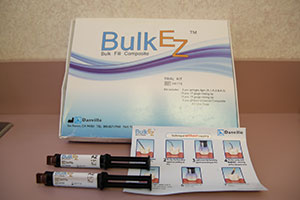
Dr. Glazer: Yes, several have come true. I envisioned a composite resin that was self-etch, self-adhesive, dual-cure to a minimum of 4.0 mm, abrasive resistant, plaque resistant, color stable, polishes to a high gloss, and is bioactive. Bulk EZ Bulk Fill Composite (Danville Engineering/Zest Anchors) (Figure 1) most closely fits these requirements. With a 4.0-plus mm depth of cure and no layering required, Bulk EZ, thus far, has demonstrated no marginal breakdown or leakage and is highly radiopaque.
I also envisioned the further advancement of laser systems for periodontal treatment and restorative procedures. When it comes to soft-tissue diode lasers, there have been several in the marketplace that have become mainstream: Picasso and Picasso Lite (AMD LASERS) (Figure 2) to name but a couple. However, hard-tissue lasers have been slow to gain market traction and this is mostly due to the cost. AMD LASERS has again broken the price barrier with the introduction of the Picasso Lite Touch all-tissue (ie, hard- and soft-tissue) laser.
 |
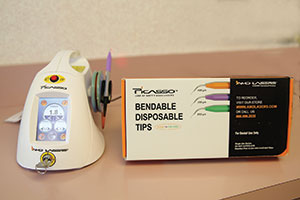 |
| Figure 1. Bulk EZ Kit from Danville Engineering/Zest Anchors. With syringes and clear photo instructions, this material can be used to bulk-fill at depths greater than 4.0 mm. | Figure 2. AMD LASERS’ Picasso Laser with Bendable Disposable Tips in assorted sizes. |
 |
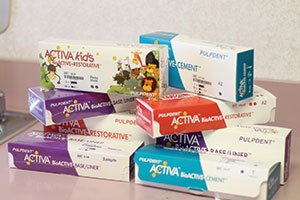 |
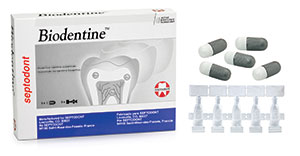
| Figure 3. Biodentine (Septodont) is a base/liner that stimulates secondary dentine growth. | Figure 4. (a) Activa BioActive Base/Liner and Activa BioActive Restorative along with Activa’s BioActive Restorative for Children (Pulpdent). All of these materials feature replacement of calcium and phosphate that is lost in the decay process. (b) Activa BioActive Cement (Pulpdent) is ideal for creating a crystalline “lock” with the tooth structure. |
 |
 |
| Figure 5. TheraCal (BISCO Dental Products) is indicated for both indirect and direct pulp capping. | Figure 6. PerioPatch (left) and PeriRinse (right) (IZUN Pharmaceuticals) both aid in reducing periodontal inflammation and promote healing. |
 |
 |
| Figure 7. Enamel regrowth is a key property of this easy patient-applied product called Remin Pro (VOCO America). | Figure 8. MI Paste Plus (GC America) can effectively minimize sensitivity and maintain healthy teeth with Recaldent’s amorphous calcium phosphate-caseinphosphopeptide combination. |
 |
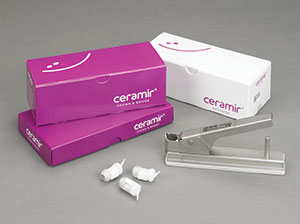 |
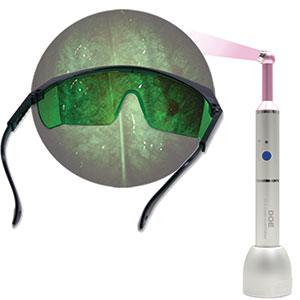
| Figure 9. The DOE system (DentLight) is an essential tool in the early detection of oral cancer. | Figure 10. Ceramir cement (Doxa) is now available in a Quick Cap with more cement per capsule and easier dispensing. |
I wished for medications that would prevent decay, regrow enamel, and maximize gingival health. There are many products in progress, but some current examples that come to mind to help prevent further decay and stimulate secondary dentin growth are Biodentine (Septodont) (Figure 3), Activa BioActive Base/Liner and Activa BioActive Restorative (Pulpdent) (Figure 4), and TheraCal (BISCO Dental Products) (Figure 5). In the periodontal arena, there are PerioPatch and PeriRinse from IZUN Pharmaceuticals (Figure 6), which both reduce periodontal inflammation and promote tissue healing after debridement of the periodontal pocket. Of course, we also have some good products to promote enamel regrowth like Remin Pro (VOCO America) (Figure 7), MI Paste Plus (GC America) (Figure 8), and Sensodyne’s ProNamel Toothpaste (GSK).
Lastly, continued advancement in early caries detection and oral cancer was one of my stated wishes. Toward that end, we have seen the development of the CariVu (Dexis), the DOE system (DentLight) (Figure 9), and the Bio/Screen Oral Exam Light (AdDent).
What do you see as the greatest change since our last interview?
Dr. Glazer: We are seeing many more bioactive materials being developed and introduced. The concept of minimally invasive dentistry has been well established, and now we are focusing on being true restorative dentists who are not simply repairing and filling holes in teeth. The value of glass ionomer (GI) and resin-modified GI materials like those from GC America and SDI that have been in the vanguard of early bioactivity with the release of fluoride led the way for bioactivity. Now we are adding minerals like calcium and phosphate to truly help restore the tooth.
You predicted bioactive materials to have an impact in restorative dentistry. Do you still feel the same way?
Dr. Glazer: What was started with Ceramir cement (Doxa) (Figure 10)—which has calcium aluminate to form a nanocrystalline lock with the tooth structure—and the use of Recaldent in MI Paste and MI Paste Plus (Figure 8) has blossomed. Today, we have bioactive restoratives that release both calcium and phosphate along with fluoride. Look no further than Activa Base/Liner and Activa Restorative (Figure 4a), which are changing the way we think about restoring tooth structure. No longer are we simply repairing a tooth. With these materials, we can truly restore a tooth by healing the tooth structure from within by replacing the calcium and phosphate lost to the decay process.
So where do you see us heading in the near future?
Dr. Glazer: As you know, I do not have a crystal ball, but I can wish for the development of something that will completely eradicate tooth decay. In a large portion of the world, we are fortunate that decay can be to some extent avoided, managed, and treated. However, like any other disease, it can have a disastrous effect in countries that have lower socioeconomic demographics. We cannot rely on fluoride alone to prevent decay, so we must continue to better educate patients on good oral hygiene, diet, and regular visits to the dentist.
What do you see as the latest and most significant material and technological advances now available to the clinician?
Dr. Glazer: As I mentioned earlier, our focus must be on prevention and true reparative materials that are bioactive, be they restoratives or cements. For example, Ceramir cement may have been the first, but now we also have Pulpdent’s Activa BioActive Cement (Figure 4b) and BioCem (NuSmile).
For technological advances, I would point to the area of anesthesia. I have long been an advocate for painless anesthesia both as a provider and a patient. In fact, I have coined the term ouchless injection (trademark pending) and, to that end, I will share the components used in my office.
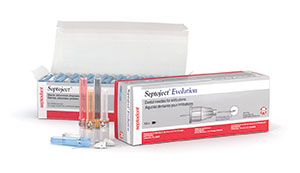
First are anesthetic needles such as Septoject Evolution (Septodont) (Figure 11) and Painless Steel (Transcodent). In both cases, these are tri-beveled so they slice into tissue, not traumatically prick the tissue. And they have siliconized cannulas to prevent tissue invagination. I prefer to buffer my lidocaine solution with 8.4% sodium bicarbonate using the cost-effective and simply unique Anutra Local Anesthetic Delivery System (Anutra Medical) (Figure 12a). Buffering makes the anesthetic more efficient and provides for faster uptake, thus making it able to achieve pulpal anesthesia in an inferior alveolar nerve block in 2 minutes or fewer. For single-tooth anesthesia, I use a modified periodontal ligament (PDL) technique with the Numbee syringe tip (Novoject) (Figure 12b). This is not a needle, but rather a cannula that is surrounded by a silicone-like material that allows the clinician to easily slip the tip into the sulcus right up to, but not penetrating, the PDL. Then you simply infuse the tissue with the anesthetic solution. The unique Numbee tip can be used with an N-Tralig (Miltex) or Citoject (Heraeus Kulzer) syringe, or any such syringe of your choosing. Lastly, I use the VibraJect R3 (Golden Dental Solutions) (Figure 12c) that clips onto the barrel of my syringe and causes a vibrating sensation to the patient. Based upon the Gate Control Theory of Pain, a nonpainful stimulus closes the gate of the pain receptors. This little snap-on device, with rechargeable batteries, is a must-have product for me with all injections.
 |
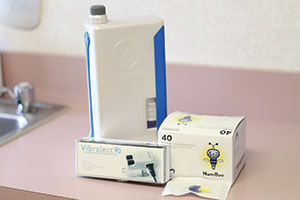 |
| Figure 11. Septoject Evolution needles (Septodont) help avoid the pain associated with the prick of the needle and invagination of the tissue by the cannula. | Figure 12. (a) The Anutra Local Anesthetic Delivery System (Anutra Medical) automatically mixes 8.4% sodium bicarbonate with lidocaine. (b) The Numbee syringe tip (Novoject) is a needleless needle for single-tooth anesthesia. (c) The VibraJect R3 (Golden Dental Solutions) minimizes any sensation of the needle piercing the tissue. |
 |
 |
| Figure 13. NoCord (Centrix) impression material has 15% alum in the wash material, thus eliminating the need for retraction cord in most cases. | Figure 14. Smiles in the Sun Seminar 2017 is a great example of how to incorporate world-class continuing dental education and an exchange of ideas with colleagues in and relaxing environment. |
 |
 |
| Figure 15. Solutionreach provides for total patient engagement via telephone, email, and text messages. | Figure 16. The ScanX Swift (Air Techniques) is a great addition to any triage, endodontic, or surgical operatory to allow for the quick processing of a single digital radiograph. |
 |
 |
| Figure 17. The EyeSpecial II (Shofu Dental) is a camera designed for dental auxiliaries to take perfect photos all with single-handed operation. | Figure 18. CamX Spectra (Air Techniques) provides a depth guide to the decay present in a tooth and captures it on your screen and in your server. |
 |
 |
| Figure 19. G-Premio Bond (GC America) is a 7th generation adhesive. | Figure 20. Adhese Pen (Ivoclar Vivadent) provides an easy and very cost-effective way to direct the adhesive resin exactly where you want it. |
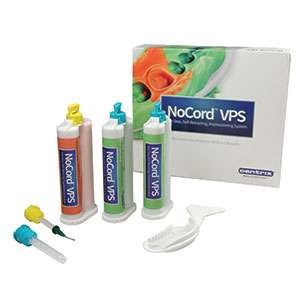
Two other technological advances are in the area of impression materials. Despite the advances in digital impressions, the cost for equipment remains high and thus most practitioners still take traditional physical impressions. Exa’lence (GC America) is a hybrid vinyl polyether silicone (VPES) material that offers flow, wettability, and high tear strength for exacting marginal detail. The second advance is an impression material called NoCord (Centrix) (Figure 13). NoCord is vinyl polysiloxane impression material that is comprised of a MegaBody tray material and a wash, and there is no retraction cord required prior to the impression. The wash material contains 15% alum to dry and open up the sulcus to allow for a great impression.
What is different in education?
Dr. Glazer: I am going to answer that in 2 ways, with regard to the professional continuing education (CE) and then with regard to patient education.
The Internet has become a focal point for people to receive information. I find this to be no less true for many of my younger colleagues. They are largely absent from major dental meetings and trade shows. Dental schools and manufacturers all offer webinars or courses almost daily (ie, dentalcetoday.com), which I believe can be a suitable form of CE. Unfortunately for those who rely on the Internet, I think they are missing a very important part of the live CE experience, and that is the social interaction of eye-to-eye conversation and camaraderie that builds valuable interprofessional friendships and mentoring opportunities, such as the 2017 Smiles in the Sun meeting in April (Figure 14).
For patients, it is important that we fully communicate with them, in a timely and clear fashion. This has fostered the use of office websites and digital communication services. The 2 that I find essential are Solutionreach (Figure 15) and Social Dental (Banyan). Solutionreach provides for total patient engagement by reaching your patients via text, email, or by recorded telephone messages. Besides the simple communication tasks of appointment reminders, recalls, and confirmations, it also sends them a monthly newsletter on various educational dental topics. Banyan’s Social Dental platform provides a unique way of communicating to current and future patients with a system of social signs and photos posted to Facebook and your website. When you realize that, statistically, the average person checks his or her smartphone almost 100 times each day, we need these systems to reach out to our patients.
How can practitioners who want to excel in dentistry differentiate themselves from all the others?
Dr. Glazer: Dr. Jack Glazer was my father and mentor, and we practiced together for nearly 14 years before he retired. He taught me, amongst many other things, that success is really spelled “s-u-c-c-c-e-s-s.” He explained that the 3 Cs stood for the level of “comfort, care, and concern” that we each provide to our patients and that will lead to our individual success.
It is important to have an Internet presence via a website. Do things to increase your search engine optimization (SEO) via Google or Facebook or Healthgrades. Both Solutionreach and Social Dental are excellent platforms provided by companies that understand our needs and are easy to work with to maximize your returns with minimal effort on your part. The dental practice must offer different services, such as learning and perfecting a painless injection technique. In addition, it is important to take CE courses that allow one to provide additional services like implants (both the surgical and restorative phases) and clear aligner orthodontics.
 |
 |
| Figures 21 and 22. Futurabond U (VOCO America) and ALL-BOND UNIVERSAL (BISCO Dental Products) are the leading 8th generation universal bonding agents. |
 |
 |
| Figure 23. Beautifil Flow Plus (Shofu Dental) has 2 viscosities so you can decide how much flow you want for a restorative procedure. | Figure 24. Unit dose G-ænial Sculpt (GC America) is available in a variety of shades. This composite packs and sculpts easily, is wear resistant, easy to polish, and will continue to self-polish with time. |
 |
 |
| Figure 25. Admira Fusion (VOCO America) comes in individually wrapped unit dose tips in a variety of shades. It is the first ORMOCER material that is based on silicone oxide and has no conventional monomers. | Figure 26. Aura (SDI) takes the guesswork out of shade selection and lets the tooth choose the shade for you with enamel, dentin chroma, and multichroma shade guides. |
 |
 |
| Figure 27. Venus Diamond and Venus Pearl (Heraeus Kulzer) offer lifelike results, great polishability, have 2 unique shades (HK2.5 and HK5), and are available in both syringe and preloaded tips (PLTs). | Figure 28. Shofu Dental’s Beautifil II PLTs allow for direct placement of the composite and minimize the entrapment of air in packable composites. Like most Shofu materials, it has Giomer chemistry that allows for the release and recharge of fluoride. |
 |
 |
| Figure 29. This pink composite (Beautifil II Gingiva “Pink” kit [Shofu Dental]) allows us to effectively treat patients who have lost the gingival covering of the root and now have decay. We can the appearance of gingiva by employing a range of pink shades to create a more natural smile. | Figure 30. (a) Fissurotomy Burs (SS White Burs) allow us to explore and conservatively restore suspicious pit and fissure lesions in a minimal amount of time, often without the need for a local anesthetic. (b) The Smart Bur II from SS White Burs is a low-speed latch type bur that enables us to only remove infected soft dentin and leave affected dentin while avoiding any iatrogenic pulp exposure. |
This is a 2-part question. (1) If you were going into practice today, what would you do differently? (2) What would be the absolutely essential products to have?
Dr. Glazer: First part first! I was fortunate to join my father in practice after completing a general practice residency (GPR) program. Today’s graduates need to do several things to help ensure their success in the short and long term. They should seek additional training immediately after dental school, when feasible with GPRs or Advanced Education in General Dentistry (AEGD) programs. Today’s graduate has to be familiar with the use of hard- and soft-tissue lasers, the use of implants for restoring function from single-tooth replacement to securing full-arch prostheses. The young graduate must understand CBCT technology and its uses, and be ready to implement the use of microscopes in general dental procedures; not just in endodontics. And finally, I do believe that the concept of group practice should be considered from both financial and quality of life perspectives.
With regard to must-have products, let me break it down by category:
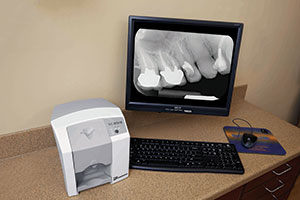
Radiography—Digital radiography is an absolute must for me, and I much prefer the more flexible and versatile phosphor plates over hard sensors. Toward that end, I use the ScanX and Swift systems (Air Techniques) (Figure 16). Specifically, I use the Swift in my triage/endo operatory. On those occasions when I do use a hard sensor, my choice is the Clio Digital X-Ray Sensors (SOTA Imaging). In my opinion, rectangular collimation is essential, and I wish more people would rally to a device such as the Tru-Image (IDIXray) that dramatically reduces “scatter” radiation.
Anesthesia—To recap what I mentioned above, it is essential to consider buffering anesthetic using the Anutra Local Anesthetic Delivery System; having the Numbee “no needle” syringe tip and the VibraJect R3 (Figure 12); and last, the anesthetic needles from Septoject (Figure 11) and Painless Steel.
Communication—One cannot overstress the importance and value of communication with patients of record and potential patients. The 2 products that fulfill those functions are, as mentioned previously, Solutionreach (Figure 15) and Social Dental.
For direct communication with the lab, case documentation, and educating the patient, I have the EyeSpecial II camera (Shofu Dental) (Figure 17). First, and foremost, is to understand that this is not a camera for the doctor. It has been specifically designed for the staff; ie, dental assistant and/or hygienist. My favorite function of this camera is the shade selection mode that will help with communicate color/shade information to the dental laboratory team. The fact that it is hygienic, lightweight, and easy to use makes this an excellent camera system.
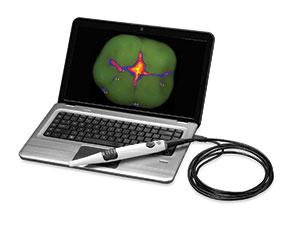
Caries detection—There are 3 systems that have impressed me with their technology: CamX Spectra (Air Techniques) (Figure 18), CariVu, and the Canary System (Quantum Dental Technologies).
Restorative materials and products—Wow! This could be an interview or article unto itself, and it seems that the changes in this category happen almost daily. For example, with adhesives, it was not long ago that we took a leap of faith from 4th, 5th, and 6th generation adhesive resins to the now popular 7th generation materials like BeautiBond (Shofu Dental), iBOND SE (Heraeus Kulzer), G-Premio BOND (GC America) (Figure 19), Peak (Ultradent Products), and Adhese Pen (Ivoclar Vivadent) (Figure 20). Now there are the universal, or 8th generation, with products like Futurabond U (VOCO America) (Figure 21), and ALL-BOND UNIVERSAL (BISCO Dental Products) (Figure 22).
When it comes to composites, the bioactive materials—such as Activa Base/Liner and Activa Restorative—can restore the calcium and phosphate lost in the decay process. For flowable resins, Beautifil Flow Plus F00 and F03 (Shofu Dental) (Figure 23), Wave hv (SDI), and G-ænial Flo (GC America) are important restoratives.
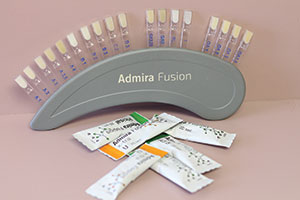
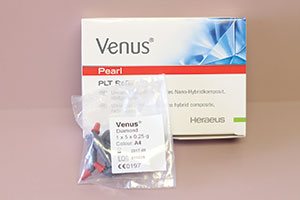
For packable composite resins, there are several that I employ to satisfy the range of aesthetic needs those patients present. Such materials, in no particular order, are: G-ænial Sculpt (GC America) (Figure 24), Admira Fusion (VOCO America) (Figure 25), Aura (SDI) (Figure 26), Venus Diamond and Venus Pearl (Heraeus Kulzer) (Figure 27), and Beautifil II (Shofu Dental) (Figure 28). For packable bulk-fill composites, I tip my hat to Tetric Evo Ceram (Ivoclar Vivadent) and Beautifil II.
 |
 |
| Figure 31. (a) The OptraSculpt Pad (Ivoclar Vivadent) was designed to eliminate the problem of composites sticking to placement instruments. (b) Composites (all manufacturers) will not stick to the disposable pads, and the resulting surface of the restoration will be 95% finished and polished. |
 |
 |
| Figure 32. The Uveneer system (Ultradent Products) employs reusable and autoclavable templates that can be used with any composite to create an immediate veneer. | Figure 33. The Fusion 5 (DentLight) is a powerful LED light that has direct collimation of at least 10 mm. Also in the photo is Nano Freedom (DentLight) wireless loupe light that allows you to turn the light on and off without touching the battery pack attached to your loupes. |
 |
 |
| Figure 34. VALO Grand (Ultradent Products) is a powerful light with a broad head for photo-curing the entire occlusal surface of molars. | Figure 35. Florida Probe with Voice Works is a must-have system for the hygienist or anyone doing periodontal charting. It will train to any voice and provides and easy to see graphic that allows you and your patients to see their needs and their results. |
 |
| Figure 36. The iOptima (Bien-Air Dental) electric handpiece system provides maximum flexibility and is controlled by an Apple iPod. |
While we are on the subject of composites, I must mention Shofu Dental’s new Beautifil II Gingiva “Pink” kit (Figure 29). As our population continues to age, we quite often see more cervical lesions encompassing gingival recession. Consequently, we can now aesthetically mask the recession and cementum with a realistically colored material that also has the benefits of Shofu’s Giomer chemistry.
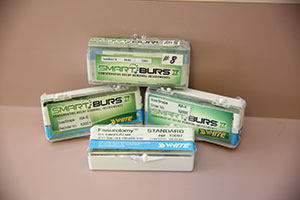
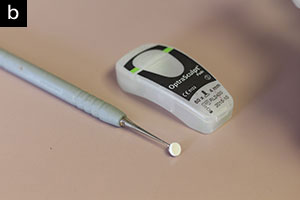
I could not restore a tooth without certain instruments. For minimally invasive decay removal I rely on SS White Burs’ Fissurotomy Burs (Figure 30a) and SmartBurs II (Figure 30b). And since all composites are sticky by nature, I use the OptraSculpt Pad instrument (Ivoclar Vivadent) (Figure 31) for placement of the composite; it will not stick to any composite and leaves the surface about 95% finished and polished. I particularly like this instrument for hand-sculpted, direct resin veneers and Class V restorations. You can be on the lookout for the introduction of a Class II tip in the near future.
For quick direct resin veneers chairside, there are 2 systems: Uveneer (Ultradent Products) (Figure 32) and the Visalys Veneer Chairside System (Kettenbach). These systems allow us to provide a very good, quick solution to aesthetic problems.
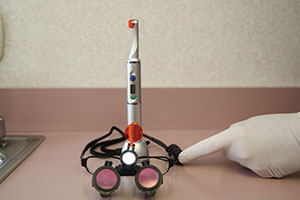
And lastly, the power of the photo-curing light you use is critical to depth of cure. I consider the 3 best curing lights in the market to be the Fusion 5 (DentLight) (Figure 33), VALO Grand (Ultradent Products) (Figure 34), and Bluephase Style (Ivoclar Vivadent).
Hygiene—While the advantages of the Florida Probe are not really new, what is new and exciting is using the Florida Probe in combination with the Voice Works headset and voice recognition software (Figure 35). Using this system allows the patient to see the results of the probing, and it provides for both education and motivation to maintain good periodontal health.
Let’s look at the 5 things you wished for nearly 2 years ago, and what your wishes for the next 5 years are.
Dr. Glazer: Mostly everything I have commented on in this interview has come to be in the last 2 to 5 years! So, with that in mind, let’s look ahead. I would like to see the following on the market: continued advancement in the use of lasers and oral cancer devices, low-dose intra- and extraoral digital radiography devices, and a simple matrix system to do a Class II MOD restoration without using multiple rings. Also, an increased use of electric drill and hard-tissue lasers to make the dental experience better for our patients. Presently, the iOptima electric handpiece system (Bien-Air Dental) (Figure 36) is the most versatile unit in the market.
How do you see dentistry being practiced in the next 5 to 10 years?
Dr. Glazer: Undoubtedly the days of the solo practitioner are numbered and we will see more and more of group practices, whether as the conventional association of 2 or more dentists or the corporate structure (such as Aspen or Heartland). Regardless of how dentistry is practiced, we must never lose sight of the fact that providing the highest quality service to the patient in a safe and efficient manner is the primary goal of patient care.
How can third-party carriers help the profession and the patient?
Dr. Glazer: Third-party carriers must keep up with modern dentistry. Our materials and procedures are now sometimes costlier and/or more time consuming, but more beneficial to patient health. Third-party carriers must understand this concept and adjust their reimbursement schedule accordingly.
Same question, but this time, how do the manufacturers help the profession and the patient?
Dr. Glazer: I honestly think that manufacturers get it; when it comes to creating dental products, they strive to make and provide us with products and technology that allow us to do the best we can for our patients. The concept of bioactive materials is fast catching on, and better instrumentation and diagnostic tools are being introduced. It is these better materials and products that allow us to optimize the clinical outcomes for our patients.
Any final thoughts?
Dr. Glazer: After 40 years in practice, I think now is the best time to be in dentistry! The products, materials, and techniques that we have today allow us to provide fantastic services for our patients and truly blend the science and art of dentistry. I must compliment Dentistry Today as a magazine that strives to provide up-to-date information relevant to the everyday practice of dentistry. And, of course, my sincere thanks to you Dr. Adams, for allowing me to offer my view on present and future materials that make dentistry faster, easier, and better for the practicing clinician, but even more so for our patients.
Howard, I want to thank you for your dedication to the profession throughout many years! I, and the entire Dentistry Today team, also want to sincerely thank you for taking the time to share your valuable knowledge and experience with our readers.
Dr. Glazer maintains a general practice in Fort Lee, NJ. He is a Fellow and past president of the AGD and a Fellow of the American and International Colleges of Dentists, American Society for Dental Aesthetics, the American Academy of Forensic Sciences, and a Diplomate of the American Board of Aesthetic Dentistry. He is a former assistant clinical professor of dentistry at the Albert Einstein College of Medicine (Bronx, NY). Additionally, he is an attending dentist at the Englewood Hospital (Englewood, NJ) and is the deputy chief forensic dental consultant to the Office of Chief Medical Examiner, City of New York. He lectures internationally and has published numerous dental articles worldwide. Currently, he writes a monthly column in AGD IMPACT entitled “What’s Hot and What’s Getting Hotter!” For the past several years, he has been named as one of the Leaders in Continuing Education by Dentistry Today. He can be reached at (201) 224-2705 or via email at hglazer264@gmail.com.
Disclosure: Dr. Glazer discloses that he was part of the development team for the OptraSculpt Pad by Ivoclar Vivadent.
Related Articles
Focus On: Third-Party Carriers
Single Crown Tooth Preparation: The 80/20 Rule
A Mid-Decade Look: What’s Hot in Materials and Instruments











