INTRODUCTION
During extraction, bone can be lost due to it being removed alongside the tooth or because of gradual bone loss over time due to the site being edentulous, as dictated by Wolff’s law.1,2 Regardless of the cause, the preservation of surrounding bone dimension and volume becomes very important for aesthetic or functional reasons, such as when trying to utilize an edentulous site for implant placement and prosthetic restoration. Traditionally, the means of preserving vertical and horizontal bone dimensions involves the use of bone grafts. These grafts could be from self (autogenous), from others (allogenic), synthetic (alloplastic), or from various animal sources (ie, porcine or bovine xenografts).1,3,4 Human bone autografts are obtained from, but not limited to, the iliac crest, mandibular ramus, or ribs.4,5 However, these procedures are often invasive, and, at times, there is not sufficient material available for grafting.3 Furthermore, although the safety and biocompatibility of these grafts have been established, many patients do not accept allografts and xenografts due to religious, social, and personal reasons.3,6 As such, researchers and clinicians have been searching for alternative sources of bone graft material for ridge augmentation and preservation, thus leading to the utilization of natural tooth structure, such as dentin.
Being that dentin and bone both are derived from neural crest cells and express the same or similar growth factors within the same family, such as TGF-Beta and BMP,7,8 it is not surprising that the ability of dentin grafts as both osteoinductive and osteoconductive agent in bone augmentation has been investigated. Therefore, even though the exact concentrations have not yet been determined, autogenous dentin grafts, similar to autogenous bone grafts, contain growth factors that may aid in a more rapid healing process and an earlier and greater acquisition of bone.3,7,8 Furthermore, the majority of teeth are discarded after extraction. As such, creating dentin particulate grafts from extracted teeth that would otherwise be discarded as medical waste can provide us with a previously unavailable source and supply of grafts that could be used for ridge preservation and augmentation.9,10
A histomorphic animal study investigating the regenerative capacity of dentin grafts saw that, compared to control groups with no graft placement, the treatment group had a significantly greater amount of bone formation as well as more mature bone being formed when evaluated at 30- and 90-day intervals. In both groups, they also saw that, compared to the 30- and 90-day samples, the amount of immature bone decreased and was replaced by mature bone with less connective tissue and a more robust vascular infiltration.11 In other words, as new bone increases in quantity and quality, the dentin graft resorbs and thus decreases in quantity over time.7,11-13 Another animal study showed greater amounts of osteopontin staining, a marker for osteogenesis and bone deposition, adjacent to the demineralized dentin graft sites as well as the borders of the newly forming vasculature.7 They also showed that, when compared to the control groups, the sites containing demineralized dentin grafts had earlier placement of immature and mature trabeculae, greater quantities of trabeculae at any given time point, and lower amounts of inflammation.7 As such, this study showed that demineralized bone graft could result in a greater quantity of bone and accelerate bone deposition and remodeling processes.
Numerous studies have shown dentin grafts to be effective in maintaining as well as enhancing ridge width and height following impacted third molar extractions,12,14-16 anterior tooth extractions,17 and sinus lifts,18,19 as well as in implant placement.9,10,13,16-22
The timing associated with implant placement after dentin grafting depends on the type and quantity of bone. Still, on average, they are placed at least 2 and 3 months after graft placement for the mandible and maxilla, respectively. Due to dentin’s ability to be remodeled, as well as the osteopromotive growth factors it has embedded within its matrix, dentin grafts are viewed to be both osteoinductive and osteoconductive.3,7 Furthermore, it has been shown in animal studies that dentin grafts result in faster healing and bone deposition at any given time frame as compared to control sites.7 Thus, they are effective in augmenting alveolar bone defects to allow for sufficient bone quantity and quality, which is required for maxillary implant placement after 3 to 6 months of healing post extraction.18,20,21 It should be noted that although a study has shown that dentin grafts provide a favorable environment for immediate implant placement in the mandible,10 the clinical application for early implant placement of the posterior maxilla has not been investigated previously. Therefore, in this case study, we wanted to report on the use of dentin particulate grafting for the application of early implant placement and simultaneous vertical sinus elevation in the posterior maxilla at 8 weeks post extraction and grafting.
CASE REPORT
The purpose of this proof-of-concept study was to determine the viability of early implant placement (at 8 weeks) into maxillary sites grafted with autogenous dentin particles. The practice of both early implant placement and autogenous dentin grafting for socket preservation have been well-documented in the literature. However, no studies to date have demonstrated that the combination of these strategies might be a feasible option, particularly in the posterior maxilla. This case report was designed to determine the osteopromotive potential of autogenous dentin and whether successful integration of dental implants placed at 8 weeks post socket preservation might be possible.
Methods
A periodontal and radiographic exam was performed to rule out any pretreatment inflammation, significant periodontal disease, or mobility. Upon examination, 2 implants and one natural tooth in the maxillary right quadrant (implant sites Nos. 3 and 4 and tooth No. 5) were deemed non-salvageable and planned for removal (Figure 1a) and simultaneous autogenous dentin grafting of the processed first premolar (tooth No. 5). Two percent lidocaine with 1:100,000 epinephrine was administered via local infiltration. Tooth No. 5 was elevated and delivered using a straight elevator and forceps. The failing implant prosthesis (Nos. 3 and 4) was sectioned, and implants were reverse-torqued and removed with forceps easily without complications. After tooth extraction and implant removal, tooth No. 5 was processed following the KometaBio SDG protocol,18 including an additional wash with a 10% EDTA solution, and prepared for use as a graft material for sites Nos. 3 to 5 (Figure 1b). Processing the tooth involved removing any enamel and restorative materials during the cleansing process.
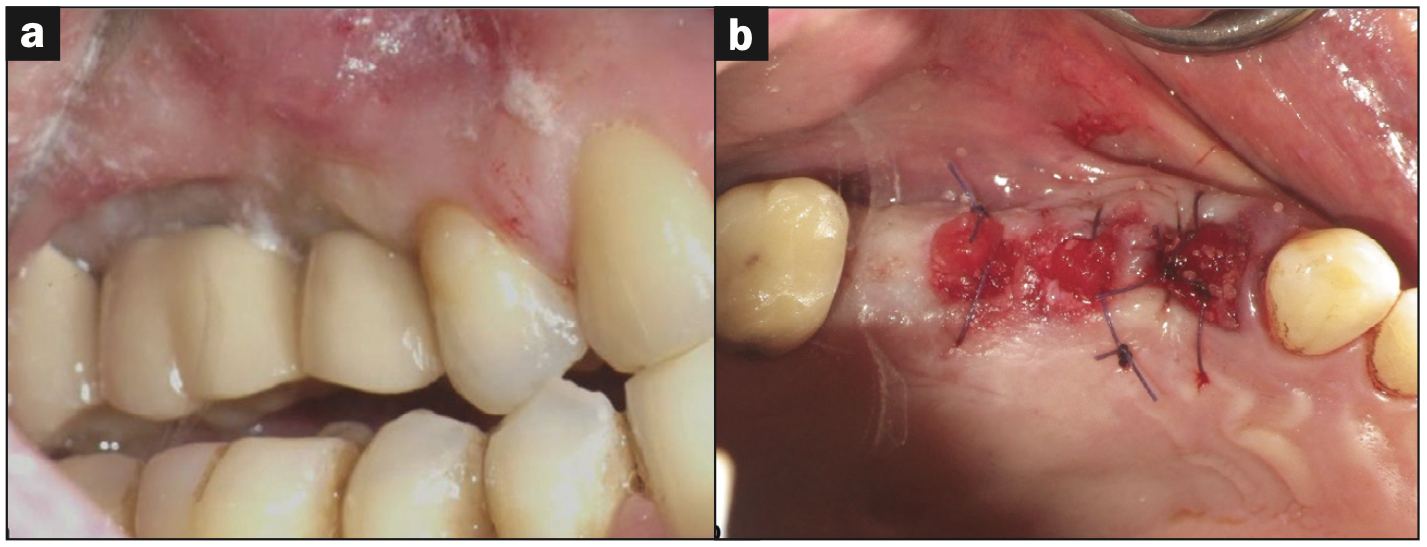
Figure 1. Extraction and grafting. (a) Preoperative evaluation of failing implants (Nos. 3 and 4) and a natural tooth (No. 5) prior to implant/tooth removal and site debridement. (b) Immediate postoperative clinical photo of ridge preservation using autogenous dentin graft. Sockets were stabilized with collaplug wound dressing and 4-0 vicryl sutures.
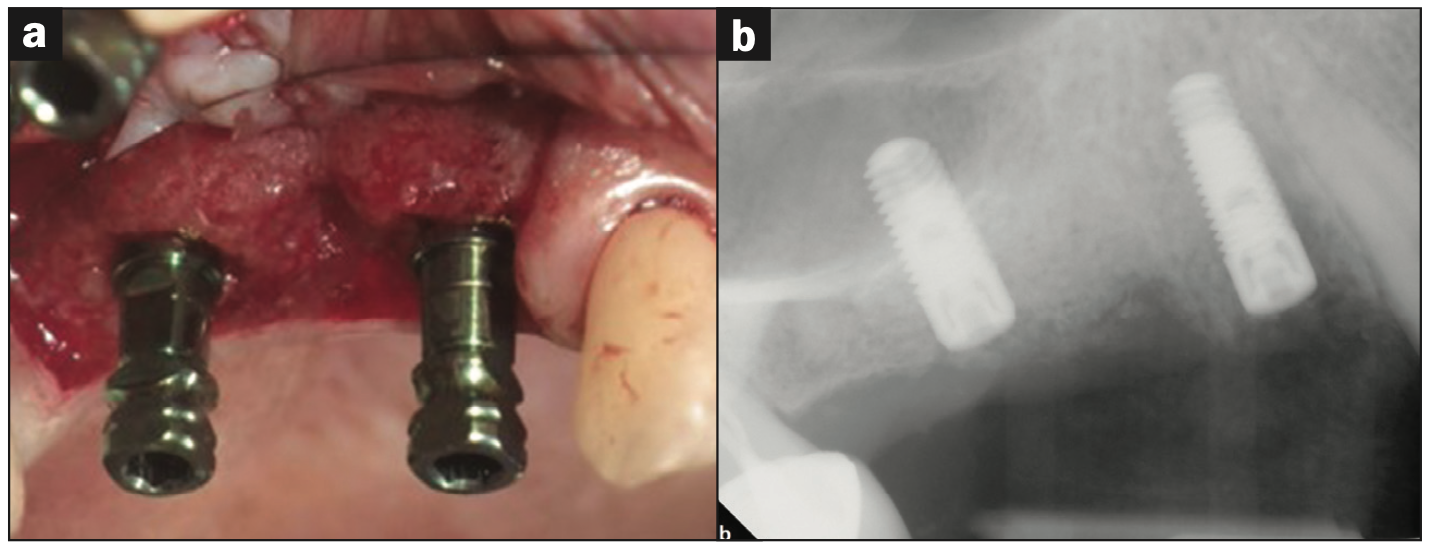
Figure 2. Biopsy and Implant Placement. (a) Second-stage surgery with flap elevation. Prior to implant placement, a core hard-tissue biopsy of site No. 5 was taken via a trephine bur. (b) Immediate post-op radiograph after implant placement with internal sinus elevation utilizing Versah protocol without complications. Primary closure was achieved with 4-0 vicryl sutures.
Re-entry surgery was performed at 8 weeks post grafting. During osteotomy preparation for site Nos. 3 and 5, a trephine core was taken from implant site No. 5 and submitted for histological analysis. Additionally, an internal sinus elevation was performed following Versah protocols without the use of additional graft material as the amount of native bone at the implant site was adequate for long-term implant stability. Implants were placed successfully with favorable insertion torques greater than 35 Ncm (Figure 2) and then successfully uncovered at 18 weeks without complications. Figures 3a and 3b denote the pre- and post-implant CBCT scans that were taken to assess healing. Overall, good primary stability of both implants was achieved and noted during the uncovery appointment.
Results
At the conclusion of the study, the clinical evaluation revealed that there was no mobility or significant inflammation present at the implant sites, and radiographically, there was no apparent evidence of significant bone loss or active disease. Based on the CBCT analysis, direct bone-to-implant contact can be noted around both implants, while an apparent increase in bone fill was noted within the maxillary sinus around the apical area of implant No. 3 due to the internal sinus elevation and subsequent fill from the osteoid-dentin complex (Figures 3c and 3d).
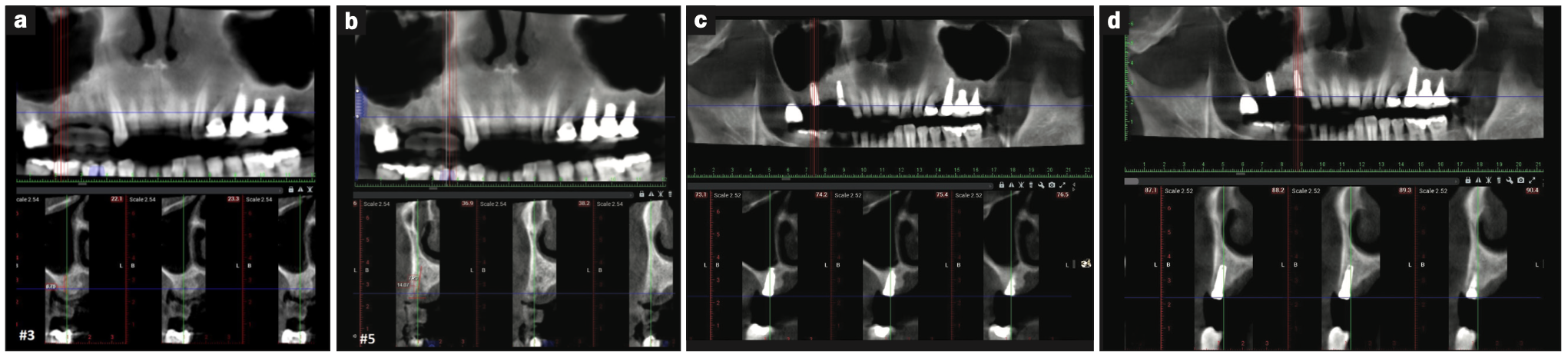
Figure 3. The 8-week post-op radiograph and CBCT images of radiographically dense surgical sites (a) No. 3 and (b) No. 5. (c and d) The 4.5-month post-implant-placement radiograph and CBCT during the uncovery appointment with good osseointegration noted.
Histologically, we noted viable new bone formation in continuity with the dentin complex, with regions of immature woven bone giving rise to mature lamellar bone (Figure 4). The implant-related inflammatory reaction was generally minimal and not interpreted to be adverse.
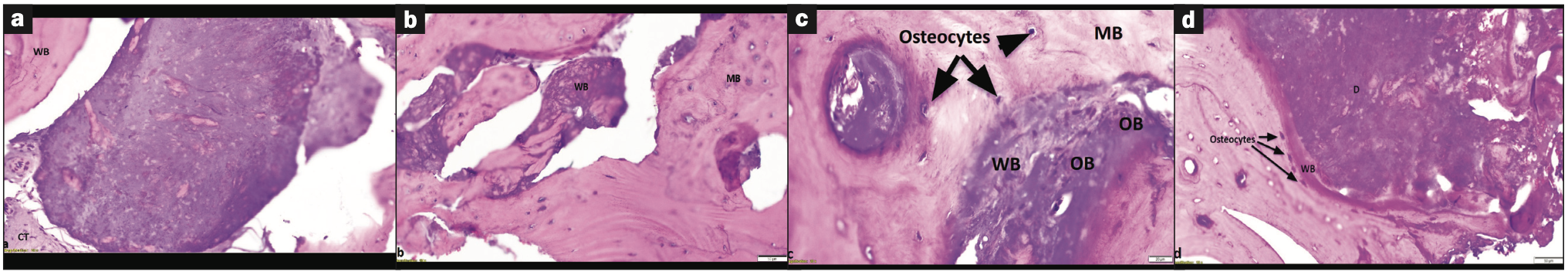
Figure 4. H&E-stained histological samples. (a) Histological image of the osteoid-dentin complex, with the core (centered) and surrounded in a background of connective tissue (CT) and the formation of adjacent woven bone (WB) (10x magnification). (b) Mature bone (MB) forming from woven bone (WB) (10x magnification). (c) Osteocytes adjacent to newly formed woven bone (WB) arising from the osteoid-dentin complex (D) (20x magnification). (d) Histological image of osteoblasts (OB) adjacent to woven bone (WB) and entrapped in mature bone (MB) formation, giving rise to the presence of osteocytes (10x magnification).
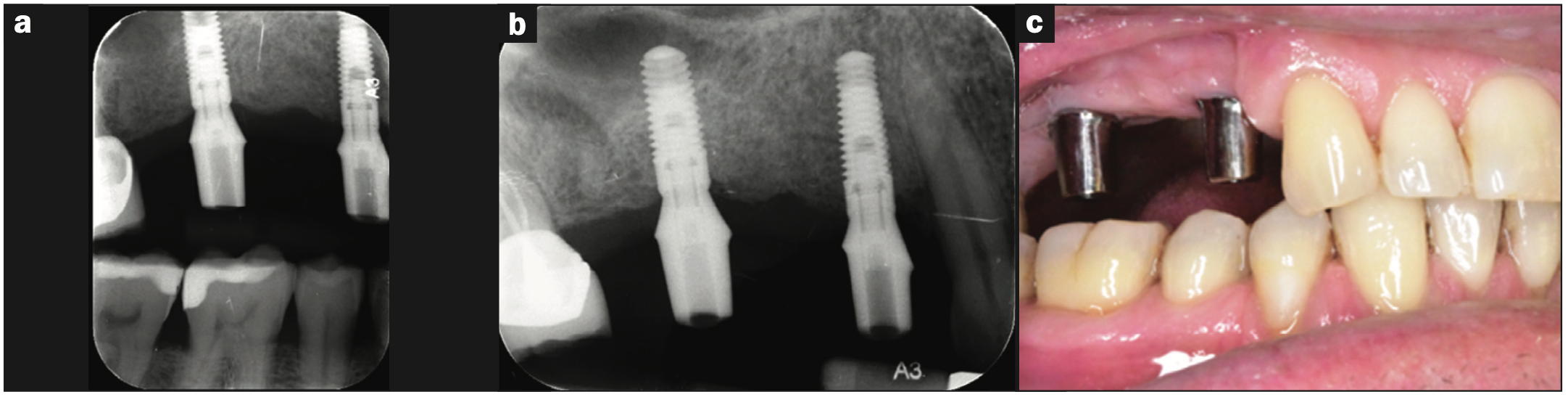
Figure 5. Clinical try-in radiographs depicting the various stages of FPD fabrication. (a) Vertical bite-wing and (b) periapical radiographs of the custom-milled abutments. (c) Buccal view of the custom-milled abutments.
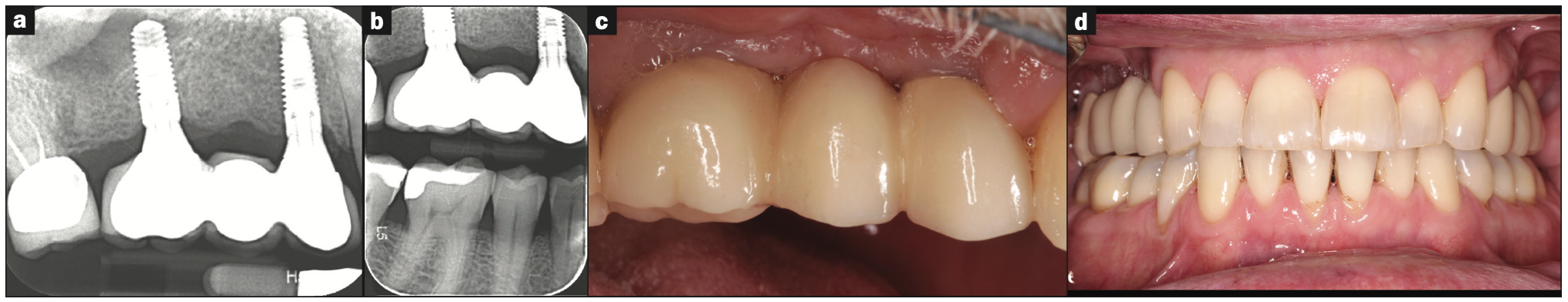
Figure 6. (a and b) Periapical and vertical bite-wing radiographs of the 3-unit FPD framework with porcelain during the delivery appointment. (c) Direct buccal and (d) facial views of FPD with porcelain added during the delivery appointment.
The 3-unit FPD was produced over 3 appointments: The first appointment involved the clinical try-in of 2 custom-milled titanium abutments on sites Nos. 3 and 5, with the buccal and palatal margins being 1 mm subgingival and flush with the gingival margin, respectively (Figure 5). The second appointment involved the clinical try-in of the metal framework prior to porcelain application. It was decided that porcelain shade A3 would be applied to the cervical and middle thirds, while a B3 porcelain shade would be applied to the incisal thirds of the prosthetic teeth. The third appointment was the clinical try-in and delivery of the prosthesis with porcelain added (Figure 6).
DISCUSSION
The lack of clinical mobility and CBCT imaging showed that there was sufficient primary and secondary stability to allow for clinically acceptable osseointegration of the implant and a successful internal sinus elevation following early implant placement into sites grafted with autogenous dentin. The histology taken 8 weeks postoperatively showed newly formed bone alongside osteocytes flanking and integrating with the dentin complex, which supports the idea that the dentin graft was ankylosed with newly formed bone. Over time, this osteoid-dentin complex will change in composition, such that the dentin will resorb and be continually replaced by bone.7,11-13 It should be noted, however, that there are differences in the literature regarding the amount of dentin that is resorbed over time. Mazor et al13 stated that although there was significant deposition of new bone after 7 months, 25% of the 7-month post-op biopsy consisted of the dentin graft, while another study indicated that there was no dentin present after a 6-month healing period in almost all subjects.1 As such, dentin grafts may or may not completely resorb, even over an extended period of time, depending on the patient population and physiology. Regardless, the dentin graft becomes ankylosed to newly formed bone, as shown by our histological samples, and should gradually be replaced by bone to a greater extent if studies investigate the graft composition changes on a more prolonged duration than 8 months. Overall, our data serves as a proof of concept that autogenous dentin grafts can be successfully used in a clinical setting for ridge augmentation, internal sinus lifts, and subsequent early implant placement in the posterior maxilla 8 weeks post-op, a parameter that has not been investigated previously.
CONCLUSION
Based on the limitations of this study, the proposed treatment of ridge preservation with autogenous dentin and early implant placement at 8 weeks may be a viable therapeutic option even in traditionally slower healing sites like the posterior maxilla. The current investigation may open new avenues into the use of autogenous dentin and its osteogenic potential, especially within the maxillary sinus. Further research into the cellular and inflammatory mechanisms behind dentin remodeling and subsequent bone formation is needed to fully understand the quality and timing of healing when using these promising autogenous graft substitutes.
ACKNOWLEDGMENTS
We would like to thank Jeffrey Eskendri, DMD, for preparing and analyzing the histological samples that were used in this paper. Dr. Eskendri is an oral and maxillofacial pathologist and is currently an assistant professor in the department of pathology at the Yale School of Medicine.
REFERENCES
1. Bartee BK. Extraction site reconstruction for alveolar ridge preservation. Part 1: rationale and materials selection. J Oral Implantol. 2001;27(4):187–93. doi:10.1563/1548-
1336(2001)027<0187:ESRFAR>2.3.CO;2
2. Pelo S, Boniello R, Moro A, et al. Augmentation of the atrophic edentulous mandible by a bilateral two-step osteotomy with autogenous bone graft to place osseointegrated dental implants. Int J Oral Maxillofac Surg. 2010;39(3):227–34. doi:10.1016/j.ijom.2009.11.004
3. Khanijou M, Seriwatanachai D, Boonsiriseth K, et al. Bone graft material derived from extracted tooth: A review literature. J Oral Maxillofac Surg Med Pathol. 2019;31(1):1-7. doi:10.1016/j.ajoms.2018.07.004
4. Raghoebar GM, Louwerse C, Kalk WW, et al. Morbidity of chin bone harvesting. Clin Oral Implants Res. 2001t;12(5):503–7. doi:10.1034/j.1600-0501.2001.120511.x
5. Roche YA, Schwartz HC. The mandibular body bone (MBB) graft: an alternative source of membranous bone. J Craniomaxillofac Surg. 1993;21(5):199-201. doi:10.1016/s1010-5182(05)80481-0
6. Güngörmüs Z, Güngörmüs M. Effect of religious belief on selecting of graft materials used in oral and maxillofacial surgery. J Oral Maxillofac Surg. 2017;75(11):2347–53. doi:10.1016/j.joms.2017.07.160
7. Fernandes AM, Mauad de Abreu FA, Fernandes MLDMF, et al. Demineralized human dentin matrix as an osteoinductor in the dental socket: an experimental study in wistar rats. Int J Oral Maxillofac Implants. 2020;35(5):910–6. doi:10.11607/jomi.8279
8. Schmidt-Schultz TH, Schultz M. Intact growth factors are conserved in the extracellular matrix of ancient human bone and teeth: a storehouse for the study of human evolution in health and disease. Biol Chem. 2005;386(8):767–76. doi:10.1515/BC.2005.090
9. Binderman I, Duda M. Extracted teeth are not a biological waste anymore: they are processed and grafted back to preserve best the alveolar ridge. PSI Imp Dent. 2018;2(18):48-52.
10. Demiraqi G, Nocka E, Sulaj E, et al. Tooth graft socket augmentation and immediate implant placement case report. Alb Dent J. 2019;18(1):14-20.
11. Calvo-Guirado JL, Maté-Sánchez de Val JE, Ramos-Oltra ML, et al. The use of tooth particles as a biomaterial in post-extraction sockets. Experimental study in dogs. Dent J (Basel). 2018;6(2):12. doi:10.3390/dj6020012. Retraction in: Dent J (Basel). 2020;8(3)
12. Andrade C, Camino J, Nally M, et al. Combining autologous particulate dentin, L-PRF, and fibrinogen to create a matrix for predictable ridge preservation: a pilot clinical study. Clin Oral Investig. 2020;24(3):1151–60. doi:10.1007/s00784-019-02922-z
13. Mazor Z, Horowitz RA, Prasad H, et al. Healing dynamics following alveolar ridge preservation with autologous tooth structure. Int J Periodontics Restorative Dent. 2019;39(5):697-702. doi:10.11607/prd.4138
14. Kuperschlag A, Keršytė G, Kurtzman GM, et al. Autogenous dentin grafting of osseous defects distal to mandibular second molars after extraction of impacted third molars. Compend Contin Educ Dent. 2020;41(2):76-82; quiz 83.
15. Sánchez-Labrador L, Martín-Ares M, Ortega-Aranegui R, et al. Autogenous dentin graft in bone defects after lower third molar extraction: a split-mouth clinical trial. Materials (Basel). 2020;13(14):3090. doi:10.3390/ma13143090
16. Schwarz F, Hazar D, Becker K, et al. Efficacy of autogenous tooth roots for lateral alveolar ridge augmentation and staged implant placement. A prospective controlled clinical study. J Clin Periodontol. 2018;45(8):996-1004. doi:10.1111/jcpe.12977
17. Valdec S, Pasic P, Soltermann A, et al. Alveolar ridge preservation with autologous particulated dentin-a case series. Int J Implant Dent. 2017;3(1):12. doi:10.1186/s40729-017-0071-9
18. Binderman I, Hallel G, Nardy C, et al. A novel procedure to process extracted teeth for immediate grafting of autogenous dentin. JBR J Interdiscip Med Dent Sci. 2014;2:154. doi:10.4172/2376-032X.1000154
19. Jun SH, Ahn JS, Lee JI, et al. A prospective study on the effectiveness of newly developed autogenous tooth bone graft material for sinus bone graft procedure. J Adv Prosthodont. 2014;6(6):528–38. doi:10.4047/jap.2014.6.6.528
20. Binderman I, Hallel G, Leretter M. Alveolar ridge preservation: Particulate dentin of extracted teeth are optimal for immediate grafting of extracted site. Dentalife. 2017;4:7-12
21. Cardaropoli D, Nevins M, Schupbach P. New bone formation using an extracted tooth as a biomaterial: a case report with histologic evidence. Int J Periodontics Restorative Dent. 2019;39(2):157-163. doi:10.11607/prd.4045
22. Joshi CP, D’Lima CB, Samat UC, et al. Comparative alveolar ridge preservation using allogenous tooth graft versus free-dried bone allograft: a randomized, controlled, prospective, clinical pilot study. Contemp Clin Dent. 2017;8(2):211–7. doi:10.4103/ccd.ccd_147_17
ABOUT THE AUTHORS
Dr. Fakhrzadeh is a board-certified periodontist practicing in New Jersey. He received his DMD degree from the Boston University School of Dental Medicine in 2014. In 2017, Dr. Fakhrzadeh completed a NIH T32 postdoctoral research fellowship in regenerative medicine at the New Jersey Center for Biomaterials. Upon completion of his research fellowship, he completed his clinical residency in periodontics and implant surgery and a master’s degree in biomedical science at the Rutgers School of Dental Medicine (RSDM) and New Jersey Medical School, respectively. His clinical and research interests are focused primarily on translational research in the fields of oral implantology and regenerative dentistry. Aside from his clinical practice, Dr. Fakhrzadeh serves as a clinical/research consultant and has held adjunct faculty positions at Rutgers University. He can be reached at amirfa9@gmail.com.
Dr. Cappetta is the director of the postgraduate periodontics program at RSDM. He received his dental degree from the University of Medicine and Dentistry, now RSDM. He studied at the Boston School of Graduate Dentistry and received his certificate of advanced graduate study in periodontology. He practiced periodontics and implantology in Summit, NJ for more than 40 years. He was president of the state board of dentistry 3 times and received a special recognition award from the New Jersey Dental Association. He is a Fellow of the International College of Dentists, an Associate Fellow in the Academy of Implantology, and a Fellow and Diplomate of the International Congress of Oral Implantology and is board-certified in periodontology and in the American Board of Oral Implantology. He has received the Outstanding Teaching and Mentoring award from the American Academy of Periodontology. He can be reached at cappeteg@sdm.rutgers.edu.
Dr. Oryan is a recent graduate of RSDM and is currently in his first year as a postdoctoral student in the department of periodontics at RSDM. He can be reached at ao419@sdm.rutgers.edu.
Dr. Drew is a professor, a director of implantology, and the vice chairman in the department of periodontics at RSDM. He received his doctorate and degree in periodontics from RSDM. He has been awarded the RSDM Excellence in Teaching Award and the Stuart D. Cook Master Educators Guild Award, and he received the prestigious American Academy of Periodontology Educator Award. Dr. Drew was inducted into the American College of Dentists, and he was awarded the RSDM Alumni Association Decade (1980s) Award. He has authored more than 30 publications and has lectured throughout the country. He was in full-time clinical practice for more than 25 years. He can be reached at drewhj@sdm.rutgers.edu.
Disclosure: The authors report no disclosures.












