While still considered a new technology, the use of lasers in dentistry actually began in the late 1980s. A dentist who is considering the clinical use of a laser in practice should consider the following questions:
• What are specific procedures that the laser would perform in my practice?
• Can one device perform all indicated clinical procedures?
• Is the technology reliable?
• Is it difficult to learn and operate a dental laser device?
• Is the substantial investment, in terms of equipment and training, justified?
This paper will provide information to help answer these questions.
LASER PHYSICS
The word laser is an acronym for Light Amplification by Stimulated Emission of Radiation. The scientific rationale for the use of lasers in dentistry can be reviewed in that context.
Light is a form of energy that travels in a wave and exists as a particle.1 This particle is called a photon. A wave of photons has 3 basic properties:
(1) A constant velocity (the speed of light).
(2) Amplitude (the vertical measurement of the height of the wave, from top to bottom). This is a measurement of energy of that wave, expressed as a joule, or 1 unit of energy. In dental applications, a useful quantity is a millijoule, one thousandth of a joule.
(3) Wavelength (the horizontal distance between any 2 corresponding points of the wave).
Both ordinary light and laser light consist of waves. However, laser light is distinguished from ordinary light by the following 2 properties:
(1) Laser light is generated as only 1 color, a property called monochomaticism. This color can be either visible or invisible to the human eye, but is described as the measurement of the wavelength, which, for dental lasers, is expressed in nanometers (one billionth of a meter).
(2) The waves of laser light are coherent for all lasers. Each wave is identical in physical size and shape.
This monochromatic, coherent wave of light energy emerges from the laser device as a precise, collimated beam. These properties make the laser beam a uniquely efficient source of energy.2
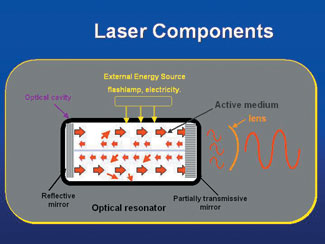 |
| Figure 1. The main components of a laser instrument. |
 |
| Figure 2. The chassis of an Erbium:YAG laser. |
 |
| Figure 3. A crystal rod of Erbium:YAG with a coin for size reference. |
Amplification by stimulated emission of radiation is a process that occurs inside the device (Figures 1 and 2). The chemical elements, molecules, or compounds in the core of the laser are called the active medium (Figure 3). This core is surrounded by a mechanism that supplies or “pumps” initial photon energy, either in the form of electrical current or by use of a rapid strobe lamp. These stimulated photons, as they reflect off of each other, create more stimulated photons, thus producing laser energy (light.) The energy is absorbed and emitted in the core, and with the aid of mirrors, is reflected and resonates within this chamber, and ultimately produces laser light. That light beam is then focused by and emitted from a small opening at one end of the device.
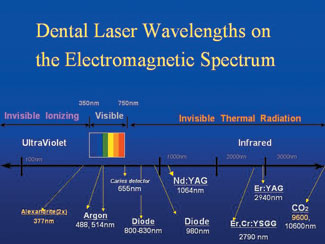 |
| Figure 4. Available dental laser instruments on the electromagnetic spectrum. Note that experimental devices are in yellow. |
Electromagnetic energy exists in waves from the smallest gamma rays, with wavelengths of about 10-12 meters, to radio waves, with wavelengths of a few thousand meters.1 Commercially available dental laser instruments all have emission wavelengths in the range of 488 nm to 10,600 nm (1 nm equals one billionth of a meter), and all are nonionizing radiation (Figure 4). This is to be distinguished from ionizing radiation, which is mutagenic as a result of effects on DNA. Dental lasers emit thermal, or heat, radiation.2,3 Four dental lasers emit light that is visible: the Argon laser, which has a 488-nm blue color and a 514-nm blue-green color; the frequency doubled Nd:YAG, which has mostly green color of 532 nm; a low-level (nonsurgical power) therapeutic device with a 635-nm red light; and another low-level caries detector with a similar red color at 655 nm.
All other laser devices (with one exception) that are employed for soft- and hard- tissue surgery emit laser light in the near, middle, and far infrared portion of the electromagnetic spectrum. The exception is one low-level laser in the range of 810 nm. The other surgical instruments are, in ascending wavelength order: 800- to 830-nm diode, in which the usual active medium consists of aluminum, gallium, and arsenate; 980-nm diode with a similar active medium; 1,064-nm Nd:YAG, where YAG is a crystal of yttrium aluminum garnet, doped (ie, coated) with neodymium; 2,790-nm Er,Cr:YSGG, which is a crystal of yttrium scandium, gallium, and garnet doped with erbium and chromium; 2,940 nm Er:YAG, where the doping agent is erbium; and 10,600-nm carbon dioxide.3
With this understanding of the physics of laser light generation, the clinician can begin the process of choosing among several devices with different features. Each practitioner should ultimately choose the device that meets the needs of his or her individual dental practice.
EMISSION MODES
Two basic emission modes are used for dental lasers: 3
• Continuous wave, meaning that the laser energy is emitted as long as the laser is activated. This produces constant tissue interaction. Carbon dioxide, argon, and diode lasers operate in this manner.
• Free-running pulse, which has very short bursts of laser energy, with each pulse being a few 10 thousandths of a second in duration. Nd and Er:YAG, as well as Er,Cr:YSGG devices operate as free-running pulsed lasers.
There are also variations of the continuous wave laser, called “gated emissions.” Gating is accomplished by mechanically closing the opening of the mirror within the laser chamber, thereby preventing laser light transmission. There is one available “super pulsed” carbon dioxide laser. This design helps minimize some of the undesirable residual thermal damage associated with continuous wave devices.
Since all lasers have a thermal effect on the target tissue, the clinician must pay attention to this effect during surgery to ensure that heat does not damage the tissue surrounding the surgical site.
DELIVE
RY SYSTEMS
Some laser instruments employ small, flexible glass fibers to deliver the energy to the tissues, while others use more rigid, tube-like devices. The shorter wavelength instruments, such as argon, diode, and Nd:YAG, have small, flexible, glass fiber optic delivery systems, with bare fibers that are usually used in contact with the target tissue. The technical challenges of conducting the longer wavelengths are quite demanding, and some manufacturers have chosen to use semiflexible hollow wave guides or rigid sectional articulated arms to deliver the laser energy to the surgical site. Some of these systems employ additional small quartz or sapphire tips that attach to the operating handpiece (a few of the devices in the erbium family); these tips may be used either in contact or out of contact with the tissues. In addition, the erbium family of dental lasers uses a water spray for hard-tissue procedures, and is the only family of dental lasers cleared for use on that hard tissue. The water is typically switched off for soft-tissue surgery. One can use an auxiliary water spray to cool the tissue when other lasers are used, but that spray is not built into the laser as it is with the erbium family.
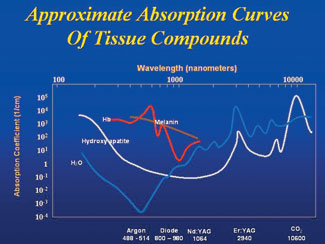 |
| Figure 5. Graph of wavelength preference for different dental tissues. |
The different wavelengths have different absorption coefficients, and this property accounts for their variable effect on human tissue (Figure 5). Water, a molecule that is universally present, is most interactive with the 2 erbium wavelengths, and somewhat less so for carbon dioxide. Conversely, the shorter wavelength lasers, including argon, diode, and Nd:YAG, have a high degree of transmission through water. Carbon dioxide, followed closely by the erbium family, is highly absorbed by the hydroxyapatite crystals of teeth and bone. There is very little interaction of the wavelengths of less than 1,000 nm with hydroxyapatite and water. However, argon, diode, and Nd:YAG do have a high affinity for hemoglobin and tissue pigments, as well as melanin. Longer wavelength laser light has little interaction with tissue pigments.
In addition to unique absorptive optical properties, all wavelengths have different depths of penetration through tissue. The erbium family of lasers is essentially absorbed on the surface of the target material, whereas the diode family can reach deeper into the tissue on the order of several thousand layers.3
It is important to emphasize that human dental hard- and soft-tissue is a combination of all of the above substances. In the treatment of dental soft tissues, the practitioner has a wide choice of devices, since all of those mentioned will have absorption by 1 or more of the tissue components. The erbium family of lasers is best for cutting hard tissues.4 The currently available continuous wave of the carbon dioxide laser produces too long an exposure time and too much heat generation for effective removal of dental hard tissues; the very short free-running pulsed erbium lasers easily ablate calcified tissue with minimal thermal effects.
LASER SAFETY
Numerous safety regulations govern the operation of lasers used in health care. Among the most important are the presence of a designated safety officer; an environment with limited access and minimal reflective surfaces; the wearing of wavelength specific protective eyewear for the surgical team, the patient, and any observer; high-volume evacuation of the laser plume; and normal adherence to infection control practices.5
 |
| Figure 6. The relationship between temperature and tissue response. |
With this background and understanding, the clinician will be able treat a variety of dental problems using different lasers. An underlying principle is the use of the least amount of energy to accomplish the treatment objective.6 The primary interaction of dental lasers with oral tissues is photothermal, ie, laser light is absorbed and transformed into heat (Figure 6). At 100C, the inter- and intra-cellular water boils, causing either soft-tissue ablation or explosive expansion and disruption of hard tissue. If the laser energy continues to be absorbed by the tissue, carbonization occurs and with it the possibility of significant tissue damage. Clearly, both target and nontarget tissues are subjected to these harmful effects. Successful treatment depends upon proper monitoring of the energy delivered, beam diameter, and duration of exposure.
There are numerous benefits of using dental lasers, but chief among them is the selective and precise interaction with the tissues. Other advantages include the ability to reduce the bacterial load (and other pathogens) in the surgical field, and, in the case of soft-tissue procedures, good hemostasis with the reduced need for sutures.7
The disadvantages of the currently available dental lasers are the relatively high cost, the need for training, the end cutting nature of the design, and the fact that no single wavelength is ideal for all clinical problems. One additional drawback of the erbium family of lasers is their inability to remove existing metallic and cast porcelain restorations.
EXAMPLES OF CLINICAL PROCEDURES
Soft-tissue excisions are easily performed with lasers. The targeted lesion is grasped with forceps or a similar instrument, or a suture, and the laser beam is directed toward a border of healthy tissue peripheral to the lesion. Keeping in mind that the rate of ablation could vary with the character of the tissue at the surgical site, the clinician should be careful not to tear any structures, but rather allow the laser energy to do the work. Sutures to close the wound are not usually necessary, and the wound will heal by secondary intention.7
Lasers are also useful for soft-tissue retraction and removal as an aspect of restorative dentistry and implant dentistry. However, the absorption characteristics of different lasers, and depth of penetration, play significant roles in treatment planning. As examples, a number of prescription medications (eg, phenytoin, cyclosporine) can cause fibrous gingival overgrowth, and the removal of that tissue can be accomplished by using the shorter wavelength lasers, which have minimal effects on enamel and cementum. Conversely, removal of the gingival tissue to uncover an implant would be best accomplished with the longer wavelength devices, ie, erbium and carbon dioxide lasers, since their energy is essentially absorbed on or near the surface, which would prevent or at least minimize heat buildup and transfer to the metallic implant fixture.8
Crown lengthening procedures, either for aesthetic improvement or for exposure of additional tooth structure for restoration placement, also can be accomplished with a laser. The argon, diode, and Nd:YAG instruments can easily remove soft tissue with very little interaction with mineralized tissue, and the erbium family of lasers can be used to expose additional root st
ructure. The tissue removal must be carefully planned so that biological width is maintained. When hard tissue is removed with the erbium instruments, the water spray must be used.
Some laser instruments have been used for removal of sulcular epithelium as part of the initial treatment of periodontal disease. This procedure is accomplished in conjunction with normal scaling and root planing, and produces a significant bacterial reduction.9,10 The laser devices employed must have a thin contact fiber that can be placed on the soft tissue lining of the pocket. The energy used would be less than one-half that employed for surgical excision.
Caries removal, tooth preparation for a restoration, and removal of defective composite filling material can be accomplished with the erbium family of lasers.11 Analyzing the absorption coefficient of the wavelengths indicates that the more available OH (hydroxyl) groups present, the more efficient the ablation. Carious tooth structure has a higher water content than healthy enamel or dentin, so the laser would be able to interact selectively with the caries. On the other hand, if in the tooth structure fluoride ions have widely replaced hydroxyl groups, the laser energy must be increased to be effective.
With the addition of thin, flexible glass tips, erbium lasers can be used in endodontic procedures; however, the currently available end-cutting energy output cannot shape the sides of canals. The ability to reduce significantly bacterial contamination of the root canal system is an advantage of the laser, and further development will enhance the use of lasers in endodontic therapy.12
There are other dental procedures where laser energy is utilized. Curing of composite resins with the visible argon laser (emission of 488 nm) results in a deeper cure and improved physical properties of the restorative material.13 Tooth whitening agents can be accelerated by various laser wavelengths; the catalyst simply needs to interact with the light, whether visible or invisible to the human eye. One device uses a visible red beam to detect dental caries via fluorescence; a sensor detects the difference between the laser light reflected from sound versus diseased tooth structure and can translate that difference into a relative numerical value. This can aid the clinician in assessing the need for treatment.12
Using energy settings much below the threshold for surgical ablation, laser therapy has been shown to be effective in reducing inflammation and stimulating new production of fibroblasts and osteoblasts for better healing. These devices emit visible red or invisible near infrared light and operate with outputs of approximately 30 to 100 mW.14
Lasers are in development that will be used for other, new applications. For example, the frequency doubled alexandrite laser (377 nm) removes dental calculus15; the 9,600-nm carbon dioxide very short pulse laser produces enamel hardening.16
CONCLUSION
When considering the use of lasers in clinical dentistry, the practitioner must use clinical experience, receive proper training, and have familiarity with the operating characteristics of each device. Because of the variable composition of human tissue and the differing ways in which laser energies are absorbed, no single laser is appropriate for all dental applications. This article has reviewed the physics of the different types of dental lasers and the clinical applications of dental lasers.
References
1. The Photonics Dictionary. 43rd ed. Pittsfield, Mass: Laurin Publishing Co; 1997:126.
2. Meserendino LJ, Pick RM. Lasers in Dentistry. Chicago, Ill: Quintessence; 1995.
3. Manni JG. Dental Applications of Advanced Lasers — 2003. Burlington, Mass: JGM Associates; 2003.
4. Aoki A, Ishikawa I, Yamada T, et al. Comparison between Er:YAG laser and conventional technique for root caries treatment in vitro. J Dent Res. 1998;77:1404-1414.
5. American National Standards Institute: ANSI Z136.3-1996 — Safe Use of Lasers in Health Care Facilities. New York, NY: American National Standards Institute; 1996.
6. White JM, Goodis HE, Chavez EM, et al. Photothermal laser effects on intraoral soft tissue in vitro. J Dent Res. 1992;71:221-226. Abstract 9250.
7. White JM, Goodis HE, Rose CL. Use of the pulsed Nd:YAG laser for intraoral soft tissue surgery. Lasers Surg Med. 1991;11:455-461.
8. Pick RM, Pecaro BC. Use of the CO2 laser in soft tissue dental surgery. Lasers Surg Med. 1987;7:207-213.
9. Raffetto N, Gutierrez T. Lasers in periodontal therapy, a five-year retrospective. J. Calif Dent Hyg Assn. 2001;16:17-20.
10. Moritz A, Gutknecht N, Doertbudak O, et al. Bacterial reduction in periodontal pockets through irradiation with a diode laser: a pilot study. J Clin Laser Med Surg. 1997;15:33-37.
11. Rechmann P, Goldin DS, Hennig T. Er:YAG lasers in dentistry: an overview. Proc SPIE. 1998;3248:2-13.
12. Ishikawa I, Frame JW, Aoki A, eds. Lasers in Dentistry: Revolution of Dental Treatment in the New Millennium: Proceedings of the 8th International Congress on Lasers in Dentistry. Amsterdam, Holland: Elsevier Science; 2003.
13. Powell GL, Ellis R, Blankenau RJ, et al: Evaluation of argon laser and conventional light-cured composites. J Clin Laser Med Surg. 1995;13:315-317.
14. Tuner J, Hode L. Laser Therapy in Dentistry and Medicine. Grangesberg, Sweden: Prima Books; 1996.
15. Pilgrim CG, Rechmann P, Goldin DS, et al. Measurement of efficiency in calculus removal with a frequency-doubled Alexandrite laser on pigs jaws. Proc SPIE. 2000;3910:50-58.
16. Featherstone JD, Fried D, McCormack SM, et al. Effect of pulse duration and repetition rate on CO2 laser inhibition of caries progression. Proc SPIE. 1996;2672:79-87.
Dr. Coluzzi graduated from USC School of Dentistry in 1970 and has been in general practice in Redwood City, Calif, since that time. He has used lasers in his practice since early 1991. He is past president of the Academy of Laser Dentistry, a faculty member of the University of California San Francisco School of Dentistry, and the 1999 recipient of the Leon Goldman Award for Clinical Excellence from the Academy of Laser Dentistry. He is coediting a new issue of Dental Clinics of North America, and is the coeditor of Wavelengths, the Academy publication.
Disclosure: Dr. Coluzzi has no financial involvement with any laser manufacturer. He purchases all laser devices and supplies used in his practice. He conducts informational and training courses for HoyaConBio Lasers and receives an honorarium for those courses.
Dr. Goldstein graduated from the University of Pennsylvania in 1968 and has a general practice in New York City. He has used lasers in his practice since 1991. He is presently the president of the Academy of Laser Dentistry and is the former editor of Wavelengths, the Academy publication. He serves on the editorial advisory board of Dentistry Today.
Disclosure: Dr. Goldstein has no financial involvement with any laser manufacturer. He purchases all laser devices and supplies used in his practice. He conducts informational and training courses for various manufacturers and receives an honorarium for those courses.


