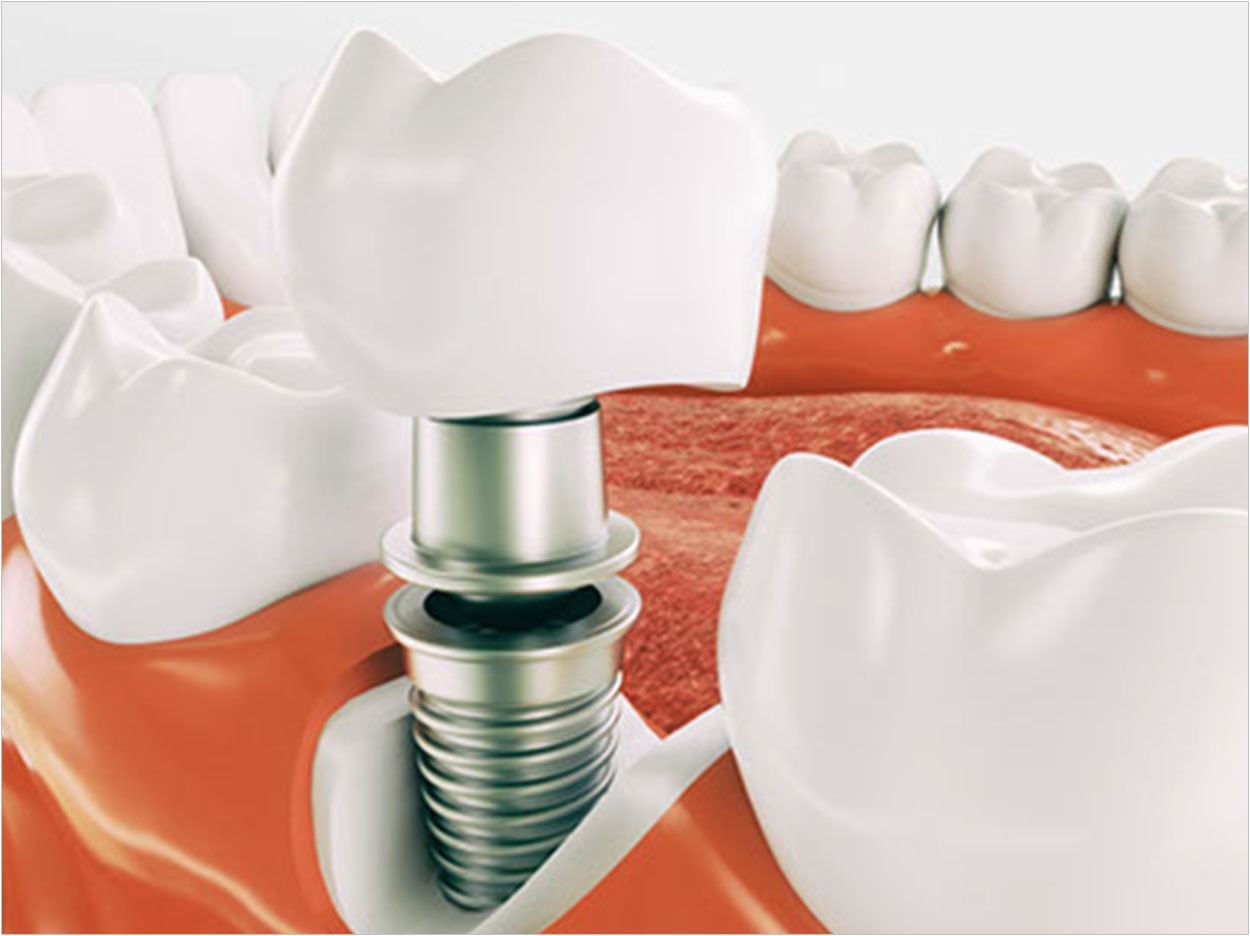
LaunchPad Medical says that it has successfully enrolled half of the 20 patients approved by the US Food and Drug Administration (FDA) for its two-site pilot clinical study, which constitutes the first clinical use of its Tetranite bone adhesive material.
The study is designed to show the safety and efficacy of Tetranite in immediately stabilizing dental implants following tooth extractions and its ability to obviate a costly, complex, and lengthy bone grafting procedure to accelerate the overall treatment timeframe for the many patients who suffer tooth loss.
“We enrolled our first patient on the first of October. The fact that we have been able to enroll half of the patients in our study in just two months indicates that this is a clinically relevant product that can address a broad patient population in the field of dentistry,” said Brian Hess, chief executive officer of LaunchPad Medical.
“I have found this material to be very easy to handle and apply,” said Michael A. Pikos, DDS, an oral surgeon and dental educator who enrolled the first patient in the study. Pikos also runs a multi-office clinical practice in the Tampa, Florida, area and is president of the Pikos Institute, which provides training in products and techniques in implant dentistry.
“Tetranite, in my opinion, has the potential to completely change our approach to many surgical procedures in dentistry, especially in the realm of implant-based surgical reconstruction,” said Pikos.
David L. Cochran, DDS, chair of the Periodontics Department at the University of Texas Health Science Center at San Antonio, leads the second clinical site in the study along with Ryushiro Sugita, DDS.
“This technology has allowed our patients to have an immediately placed implant after we extracted their tooth. All patients treated so far have tolerated the procedure well and are healing nicely. I believe that this technology will change the way dental implants are placed,” said Cochran.
Cochran also was the study director of the preclinical testing that provided the key animal safety and efficacy data that enabled the FDA to approve initiation of this pilot study, the company said. The results of the animal study will be published in the January/February issue of the International Journal of Oral and Maxillofacial Implants.
Related Articles


